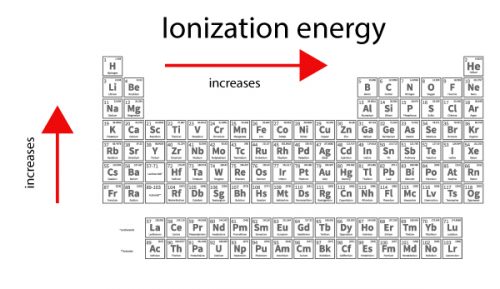
For example, the first ionization energy of boron is lower than the first ionization energy of beryllium. There are some exceptions to the ionization energy trend. But, each row adds a new shell, so the outermost electrons are still further from the nucleus. Atoms have more protons moving down a group, which does pull in the electron shells. The reason is that principal quantum number of the outermost ( valence) electron increases moving down. Ionization energy generally decreases moving from top to bottom down an element group (column).This happens because more protons are added, increasing the attraction between the nucleus and electrons and drawing the electron shells in closer. The reason is that the atomic radius tends to decrease moving across a period. Ionization energy generally increases moving from left to right across an element period (row).Ionization energy displays a trend on the periodic table. Francium, an alkali metal located on the bottom left of the table, has one of the lowest ionization energies. The element with the highest ionization energy is helium, which is located in the upper right side of the periodic table and is one of the noble gases. Ionization Energy Trend on the Periodic Table In chemistry, periodicity refers to repeating trends in the elements of the periodic table, such as ionization energy, atomic radius, electron affinity, and electronegativity. Atoms with high ionization energies (such as the noble gases) display low reactivity and are less likely to form chemical bonds and compounds. So, atoms with low ionization energies (such as alkali metals) tend to be highly reactive and readily form chemical bonds. The higher the ionization energy, the harder it is to remove an electron. Ionization energy reflects how difficult it is to remove an electron from an atom, so it is a useful predictor of reactivity and the strength of chemical bonds that atom forms. Removing each subsequent electron requires more energy. The first ionization energy is the lowest. There are exceptions to this periodic table trend.įrancium (an alkali metal) has the lowest ionization energy, while helium (a noble gas) has the highest ionization energy. Ionization energy increases moving across a period and decreases moving down a group. Ionization energy is the energy required to remove an electron from an atom or ion. Ionization energy is denoted by the symbols IE, IP, ΔH° and has units of kilojoule per mole ((kJ/mol) or electron volts (eV). The term is also spelled ionisation energy (British English). It is a periodic table trend that increases moving across the table and decreases moving down it.īy definition, ionization energy is the minimum energy needed to remove the most loosely bound electron from a gaseous atom or ion. Ionization energy is the energy required to remove an electron from an atom. 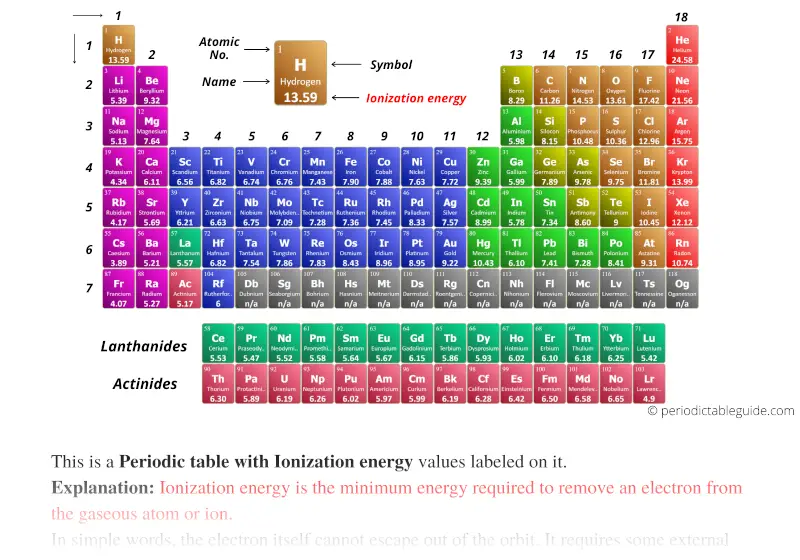
This entry was posted on Septemby Anne Helmenstine (updated on August 31, 2022)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
